|
So the molecular weight of acetic acid is 60.052. Step 4: Calculate the molecular weight of acetic acid by adding up the total weight of all atoms. the solution is 1.67 M In case of acetic acid, 1 N solution 1 M solution Thus normality of 10 acetic acid is 1.67. 
Thus 1.67 moles of acetic acid is present in 1 L of water i.e. So 100 g of acetic acid means 100/60 1.67 moles of acetic acid. The acid dimerizes in benzene due to the presence of hydrogen bonding between its molecules. 1 mole of acetic acid has a mass of 60 g. Total weight of Oxygen atoms in acetic acid: 15.9994 x 2 = 31.998 PdCl2 in acetic acid and also from LiCl and PdCl2 in the same solvent has been deduced from molecular weight and spectrophotometric data and equilibrium. Acetic acid has a molar mass of 60 g/mol.  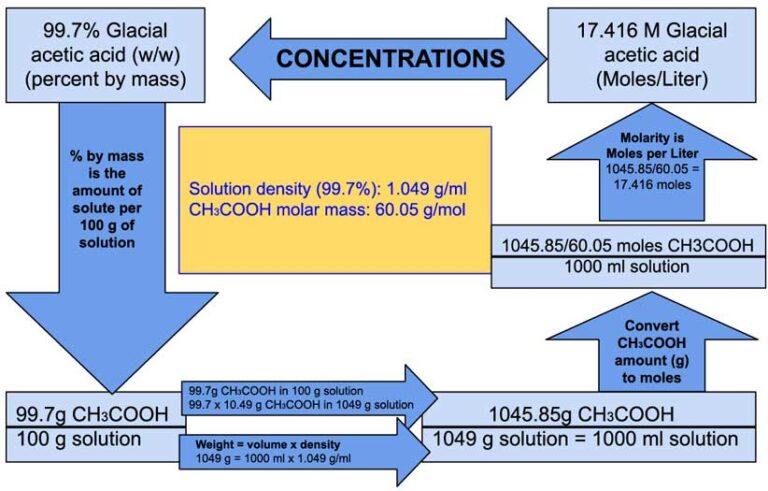
Total weight of Hydrogen atoms in acetic acid: 1.008 x 7 = 4.032 Acetic acid is one of the simplest carboxylic acids. Number of Hydrogen atoms in acetic acid: 4 Molecular weight of Acetic Acid Molar mass of CH3COOH Convert grams Acetic Acid to moles or moles Acetic Acid to grams Molecular weight calculation: 12.0107 + 1.007943 + 12.0107 + 15.9994 + 15.9994 + 1.00794 Percent composition by element Similar chemical formulas Note that all formulas are case-sensitive. Total weight of Carbon atoms in acetic acid: 12.0107 x 2 = 24.0214 Step 3: Calculate the total weight of each atom in an acetic acid molecule by multiplying its atomic weight by its number. What is the concentration of acetic acid in a solution made by dissolving 175 g of acetic acid in enough water to make 2.00 L of solution The molar mass of. Step 2: Find out the atomic weights of each atom (from the periodic table).Ītomic weight of Carbon (C): 12.0107 (Ref: Jlab-ele006)Ītomic weight of Hydrogen (H): 1.008 (Ref: Lanl-1)Ītomic weight of Oxygen (O): 15.9994 (Ref: Jlab-ele008) From the chemical formula, you can find that one molecule of acetic acid consists of two Carbon (C) atoms, four Hydrogen (H) atoms, and two Oxygen atoms. The chemical formula of acetic acid is CH 3COOH. The carbon atom has a molar mass of 12.01 g/mol, the hydrogen atom has a molar mass of 1.008 g/mol, and the oxygen atom has a molar mass of 32 g/mol. You will know different atoms and their number in an acetic acid molecule from the chemical formula. Products Chemical formula, CHCOOH Molar Mass, 60.05 g/mol HS Code, 2915 21 00 Quality Level, MQ200 Application, Acetic acid 99-100 for synthesis. Ethanoic or acetic acid has a pungent vinegar odour and sour taste. It can be calculated as: (2 × 12.011) + (4 × 1.00794) + (2×15.999) g/mol 60.05 g/mol Physical Properties of Acetic Acid Some important physical properties of acetic acid are listed below. Step 1: Find out the chemical formula and determine constituent atoms and their number in an Acetic acid molecule. By calculation, the molar mass of acetic acid comes out to be 60.05 g/mol. CALCULATION PROCEDURE: Acetic Acid (CH 3COOH) Molecular Weight Calculation The molecular weight of Ethanolamine is 60.052 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements. Do not forget to multiply the number of atoms of the element present in the compound.The molecular weight of acetic acid (CH 3COOH) is 60.052.Īcetic acid (CH 3COOH) is an organic compound of three elements: Carbon, Hydrogen, and Oxygen. 
The other name of acetic acid is Ethanoic acid because there are two carbon atoms present in it. Ammonium Acetate Formula- It is a salt that has interesting chemical properties and. Therefore, the molar mass of acetic acid is 60.05 g/ mol. Vinegar is at least 4 acetic acid by volume, making acetic acid the main component of vinegar apart from water and other trace elements. Ethanoic acid (another name) colorless liquid Unpleasant order. This online calculator you can use for computing the average molecular weight (MW) of molecules by entering the chemical formulas (for example C3H4OH(COOH)3 ). Acetic acid / sitk /, systematically named ethanoic acid / nok /, is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH (also written as CH3CO2H, C2H4O2, or HC2H3O2 ). Hint: Acetic acid is the compound that comes under the group of carboxylic acid because there is a –COOH group present in the compound and there are two carbon atoms present in it. The molar mass of acetic acid is 60.052 g/mol.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
